MISSION
To accelerate the transition to a renewable energy society by discovering new materials, chemicals, and processes through multi-scale simulation and data science.
RESEARCH
We interface multi-scale materials simulation and data science. Specifically, we develop innovative methods that accelerate materials design.
Latest Publications
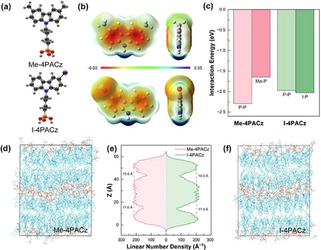
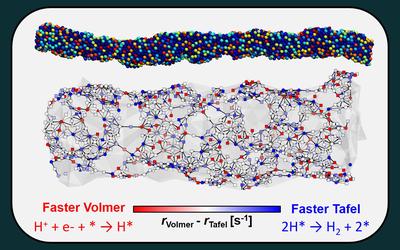
Although halide perovskite solar cells have achieved remarkable efficiencies close to their theoretical limits, durability against external factors and reproducible processing remain major barriers to commercialization. Conventional ammonium halide-based surface passivation strategies enable effective defect passivation and charge selectivity via 2D/3D heterostructure formation, but their metastability and strong sensitivity to processing conditions limit long-term stability and process window. In this work, we report an amorphous π-conjugated passivator, (4-(3-iodo-9H-carbazol-9-yl)butyl)phosphonic acid (I-4PACz). The asymmetric iodine substituent enhances the molecular dipole moment while simultaneously altering intermolecular interactions, thereby suppressing ordered molecular packing and enabling the formation of a uniform interfacial layer. The characteristics of I-4PACz result in efficient charge extraction as well as widening process window. Perovskite solar modules applied to I-4PACz as passivator between the perovskite and hole transport-layer show a power conversion efficiency of 21.2% with 24.5 cm² aperture size, and excellent long-term stability retaining 85% of the initial efficiency for 884 h under 65°C and 40% relative humidity, and 98.7% of the initial efficiency for 525 h under continuous illumination, respectively. Moreover, the minimal dependence on blade-coating speed also reinforces the suitability of this interfacial control strategy for inline and large-area manufacturing.
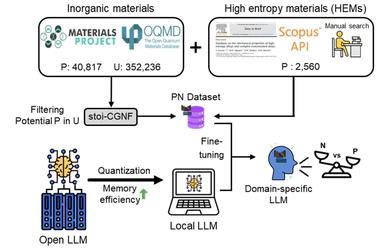
High-entropy materials (HEMs) offer unprecedented opportunities for superior mechanical, thermal, and catalytic properties, but their vast chemical space makes experimental discovery resource-intensive. State-of-the-art commercial large language models (LLMs) notably fail at HEM synthesizability prediction, a critical bottleneck in materials development. We demonstrate that domain-specific fine-tuning transforms open-weight local LLMs into accurate predictors. Using a dataset of 321,083 inorganic compositions with 2,560 HEM examples, we fine-tuned three 4-bit-quantized models (gpt-oss-20b, Qwen3-14b, and DeepSeek-R1-Distill-Qwen-14b), achieving remarkable balanced accuracy of 0.957, 0.961, and 0.956, respectively. Critically, these models operate efficiently on accessible hardware (< 15GB VRAM), eliminating costly API dependencies while ensuring data privacy and consistent reproducibility. This work could open new pathways toward autonomous closed-loop discovery, where distributed local models enable rapid screening and iterative improvement through experimental feedback. Future collaborative efforts in open data sharing, particularly including negative results, would address current fragmentation in synthesis reporting and accelerate community-wide HEM discovery.
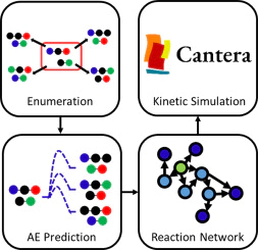
An automated rapid microkinetic simulation is invaluable in predicting the products of chemical reactions ahead of experiments. Although existing models perform well in their focused domains, a dedicated comprehensive framework for the bond exchange reaction kinetics is absent. To address this, we present an integrated algorithm for the rapid kinetic simulation of bond exchange reaction networks. We introduce a scalable matrix-based enumeration method that allows economic exploration of all plausible reaction products without resorting to reaction templates. Our model primarily uses machine learning, which achieves a mean absolute error of 4.55 kcal/mol for activation energies, to more efficiently predict reaction properties, without relying on stored chemical reaction databases or expensive electronic structure calculations. The framework was validated by successfully reproducing two reactions.
Latest News and Gallery
P.S. The cake was enjoyed by the grad students themselves. 🎂



